A study on dental instrument reprocessing management system based on wireless ICT and RFID technology
Article information
Abstract
Purpose
This study aimed to implement a dental instrument reprocessing management system based on wireless ICT and RFID technology and evaluate its operational efficiency and applicability in the dental clinical setting. Materials and
Methods
For the development of the instrument reprocessing system, hardware components including RFID tags, RFID readers, wireless internet devices, and a central server were integrated with software management programs. Once operational, the system collected information in real-time at each stage of the reprocessing workflow. System performance was evaluated in terms of stability, efficiency, and data accuracy.
Results
During pilot operation, the system automatically recorded the reprocessing history of each instrument in chronological order. Functions such as verification of sterilization completion, real-time status monitoring, error detection in sterilization, and validity management were successfully implemented. Compared to traditional paper-based recording, instances of missing records and errors were significantly reduced, and rapid traceability was enabled in case of suspected infection events.
Conclusion
The RFID-based dental instrument reprocessing management system can overcome limitations in repetitive and complex reprocessing workflows in dental practice, enhance operational efficiency, and provide tangible support for improving infection control standards.
Introduction
Medical devices reused in clinical settings must undergo thorough cleaning, disinfection, and sterilization processes to prevent cross-infection between patients [1,2]. Inadequate execution of these reprocessing procedures can increase the risk of healthcare-associated infections (HAIs) [3-5]. In particular, dental practice often involves the repeated use of small, precise instruments that come into direct contact with blood and bodily fluids [1,6], making the accuracy and consistency of sterilization processes critical for patient safety and infection prevention [7,8].
The Centers for Disease Control and Prevention (CDC) mandates the establishment of standard operating procedures (SOP) for instrument reprocessing, specifying systematic management of cleaning, sterilization, storage, and use [9]. However, in actual clinical settings, it is challenging to verify in real-time whether each process is conducted according to SOP or to systematically review the reprocessing history of individual instruments afterward. Widely used paper-based or handwritten records are prone to omissions and errors due to increased workload [10], and it is difficult to instantly confirm the sterilization status or usage history of specific instruments.
As an alternative to overcome these limitations, recent clinical settings have focused on instrument management methods utilizing Radio Frequency Identification Device (RFID) technology [11-14]. RFID is a contactless technology that uses radio waves to identify and track objects, enabling automatic recognition of instruments without individual scanning or visual inspection [15-17]. The efficiency of this technology has been validated in logistics and inventory management [18], and its potential application to the tracking and infection control of repeatedly used medical devices has been suggested [19,20].
An RFID-based sterilization and instrument management system can continuously record and manage the entire cycle, from the collection of contaminated instruments to cleaning, sterilization, storage, and reuse. This allows for rapid identification and traceability in case of issues during sterilization, enhancing reliability in the reprocessing workflow, and potentially contributing to an overall improvement in infection control standards. Nevertheless, the application of wireless ICT-based systems integrated with RFID for dental instrument reprocessing remains limited.
Therefore, this study aimed to implement a dental instrument reprocessing management system utilizing wireless communication–based ICT (information and communication technology) and RFID technologies, and to examine its operational efficiency and practical applicability by evaluating its performance through simulations conducted in a controlled laboratory environment.
Materials and Methods
The dental instrument reprocessing management system proposed in this study is an integrated system that utilizes wireless ICT and RFID technologies to automatically record and manage the entire workflow of dental instruments, including cleaning, sterilization, storage, and use.
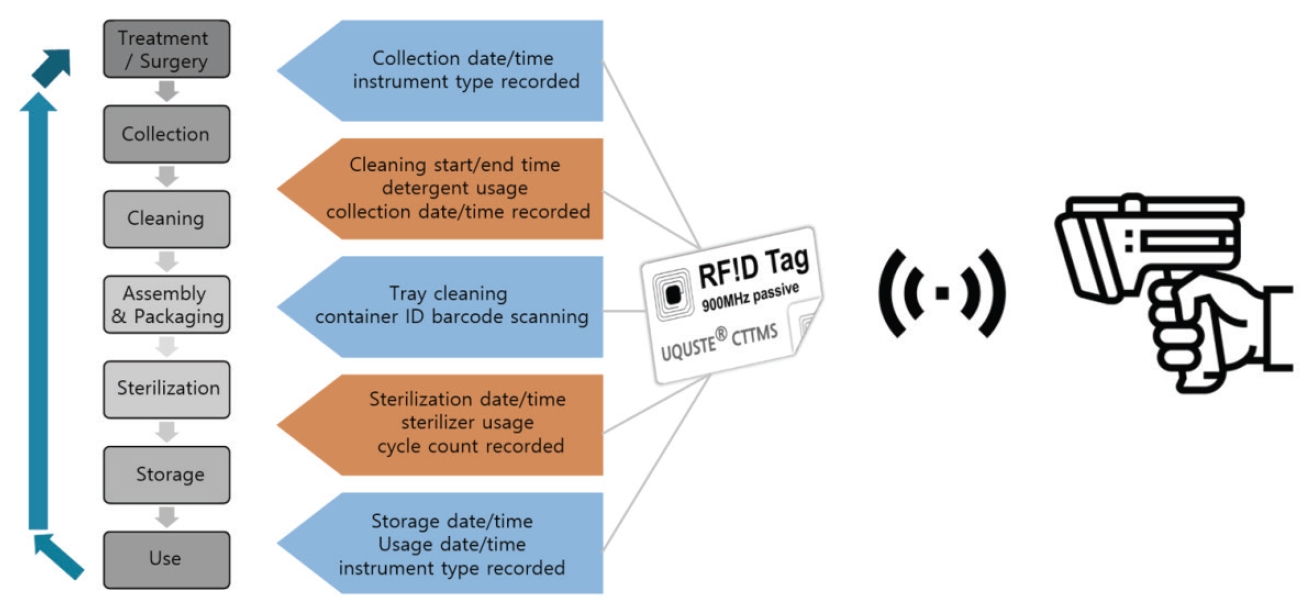
The system consists of hardware components (RFID tags, RFID readers, wireless internet devices, and server/cloud) and software (management program). All components are interconnected to enable real-time collection, transmission, and storage of information at each stage of instrument movement and processing (Fig. 1).
RFID tags attached to sterilized instruments or instrument sets identify each item based on unique identification information. Each stage of the workflow including the start and end of cleaning, storage, and actual use is automatically recognized by RFID readers. The captured data are transmitted to a central server via a wireless network, where the processing history is accumulated and stored.
RFID is a contactless technology that uses radio waves to identify objects. In this study, RFID was applied as a key component to enable automatic tracking and management of dental instrument reprocessing history.
RFID tags were attached to dental instruments or instrument sets (trays) and assigned unique identification numbers (IDs). Since most dental instruments are composed of metallic materials such as stainless steel, metal interference is a major factor affecting RFID signal performance. In this study, on-metal RFID tags (metal tags) were used to minimize metal interference. In addition, the tags were designed to withstand repeated use without physical damage or data loss under the high-temperature and high-pressure conditions of steam sterilization (autoclave) as well as the vibration and humidity conditions of ultrasonic cleaning.
Sterilized instruments were organized as sets, with RFID tags attached to allow batch recognition (Fig. 2). Small RFID tags were also attached to individual dental instruments, enabling instrument-level management (Fig. 3). Since the RFID ICs were attached to steel containers and trays, IC chips specifically designed for strong performance on metal surfaces were applied to prevent wireless communication interference. In addition, the mounting position of the IC tags was adjusted so that the metal instruments themselves could be utilized as antennas, thereby improving communication performance.

A. RFID Tag. B. RFID tag attached to the outside of the sterilized tray. C. RFID tag attached inside the sterilized tray.
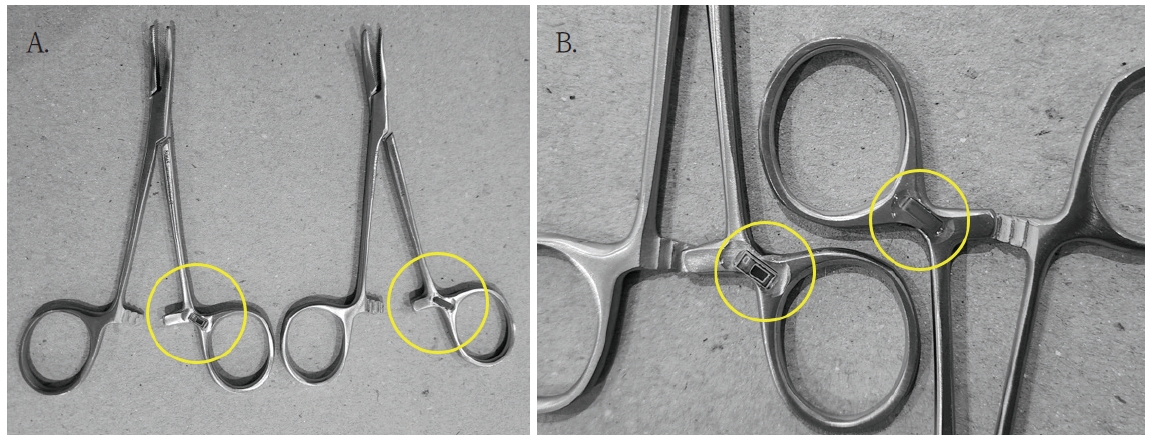
A. Small RFID chip attached to the surgical instrument. B. Enlarged view of the RFID attachment site: does not interfere with instrument use.
RFID readers detect RFID tags throughout the entire instrument movement pathway. Two types of readers can be applied fixed and mobile (handheld). The system allows not only batch scanning of instrument sets but also selective scanning of individual instruments.
By applying RFID technology, the cleaning, sterilization, storage, and usage history of sterilized instruments are automatically recorded in real time, eliminating the need for manual documentation (Fig. 4).

A. RFID chip and handheld RFID reader. B. Tagging RFID chips attached to the outside of the tray using a handheld RFID reader. C. Handheld RFID reader stored on a stand.
In this system, the wireless internet equipment is responsible for real-time data transmission between the RFID readers and the central server (or cloud server). Considering the characteristics of the dental clinic environment, a Wi-Fi-based wireless network was implemented, and access points (APs) were strategically installed in areas with frequent instrument movement, such as the central supply room, treatment rooms (operating rooms), and sterilization room, to ensure stable wireless communication (Fig. 5). The operating frequency band of the RFID tags used in this study was ultra-high frequency (UHF) at 918 MHz.

A. RFID system communication equipment. B. Spectrum Analyzer (RFID UHF tag frequency range measurement).
Through the wireless network, information is immediately transmitted to the central server. In the event of network failure, data are temporarily stored in local memory to minimize data loss and maintain continuity of instrument reprocessing records.
Moreover, the wireless network incorporates authentication-based access control and encrypted communication to enhance security, preventing leakage of patient information and sterilization history data. This wireless internet configuration allows real-time monitoring of instrument status in dental clinical settings and enables efficient infection control and instrument tracking management without location constraints.
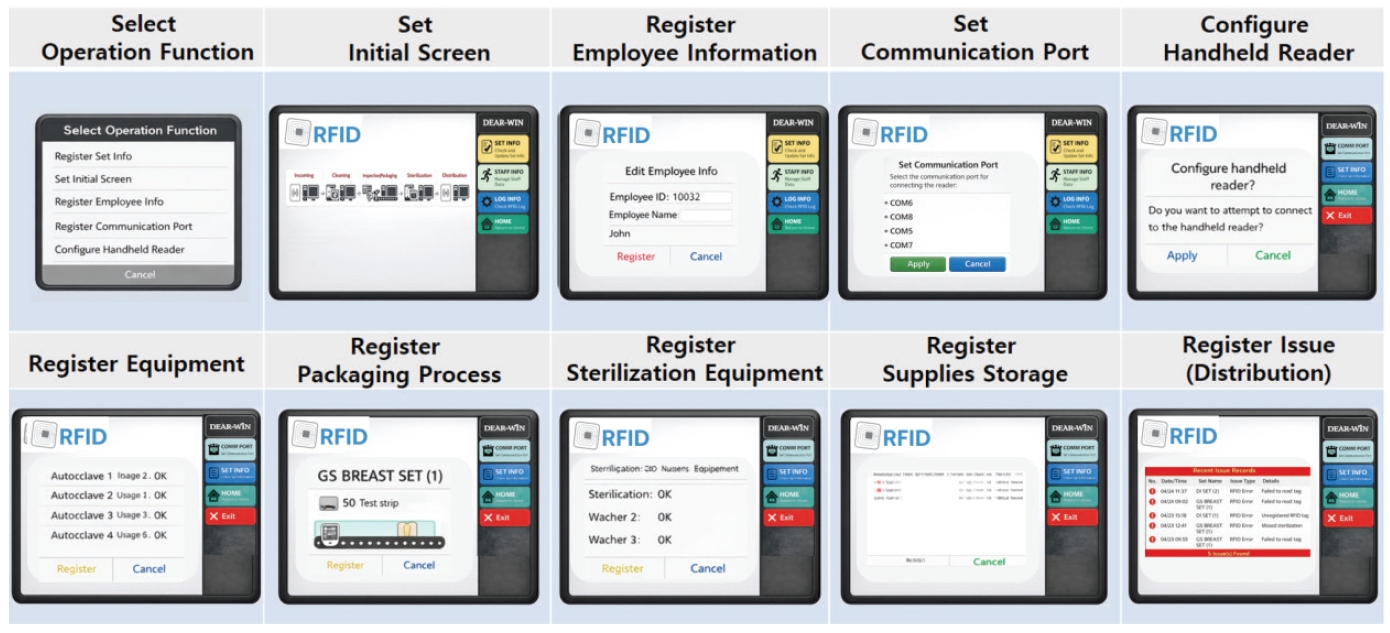
The management program (Den-win) processes data collected from the hardware and provides information according to user requirements. The status of sterilized instruments is intuitively displayed as “In Use,” “Being Washed,” “Sterilization Completed,” “In Storage,” or “Expired.” This allows medical staff and sterilization personnel to monitor instrument status in real time without manual record-keeping.
Additionally, information such as sterilization method, date and time, and personnel in charge is automatically linked and managed. The system provides administratorlevel functions, including user access control, log review, and statistical analysis. These features enable analysis of management indicators such as instrument usage frequency, time required for sterilization processes, and record omissions (Fig. 6).
In this study, we conducted simulations in a controlled laboratory environment to evaluate the performance of the dental instrument reprocessing management system developed using wireless ICT and RFID. The equipment used in the experiments is listed in Table 1.
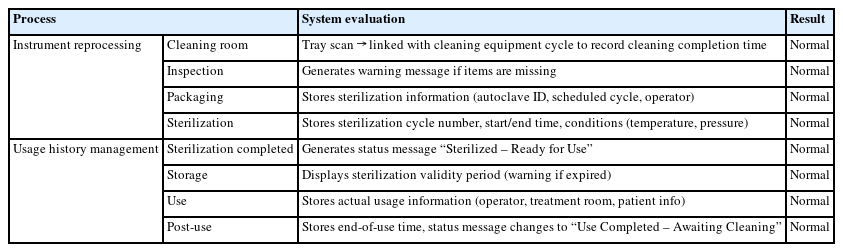
A total of six simulation runs were performed, and the cycle evaluation method was as follows. At each process step—cleaning, inspection, packaging, and sterilization— RFID tags were read to assess whether the system was operating correctly. Next, at the sterilization completion, storage management, and usage stages, RFID tags were read to verify that state changes were correctly recorded. Collected data were analyzed to confirm the accuracy of processing history and status changes, as well as to identify any missing records.
The results were evaluated in terms of system stability, operational efficiency, and information accuracy, and the records for each process step were checked for con-sistency with actual workflow. The evaluation items are listed in Table 2.
Results
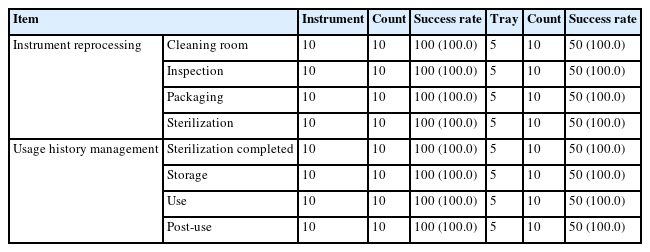
As a result of system operation, usage information was successfully visualized for all cycles, and the data were properly stored on the server. The system functionality was evaluated through 10 RFID tag readings using the reader, and all trials were completed without errors, confirming proper system operation (Table 3). The system also maintained correct functionality after 10 repeated sterilization cycles.
Upon application of the system to the instrument cleaning and sterilization processes, the status information of the instruments (Cleaning, Sterilizing, Sterilized, Stored, and In Use) was automatically updated (Fig. 7). Before clinical use, the system verified that only sterilized instruments within their valid period were available. After procedures, RFID recognition recorded the usage history of each instrument on the server. This implementation streamlined staff tasks and enabled automated and standardized tracking of instrument reprocessing (Table 4).
Furthermore, a signal interference avoidance algorithm was applied to minimize radio interference and collisions, enabling the collection of data for instrument sets without recognition errors. The installation position and angle of the antennas also played a critical role in reducing interference. Additionally, the system was designed to absorb and redistribute energy reflected from metal surfaces, thereby enhancing sensitivity.
Discussion
This study aimed to explore ways to enhance the reliability of infection control and improve workflow efficiency by systematically managing the reprocessing history of dental instruments using RFID technology.
The results demonstrated that applying RFID technology enabled the real-time automatic recording of each instrument’s cleaning, sterilization, storage, and usage history, which can improve transparency and traceability in the instrument reprocessing workflow. Healthcare facilities are required to document and retain sterilization logs and ensure the quality of sterilization processes [21-23]. However, previous studies have reported that sterilization logs are often not recorded or retained, and in cases where issues arise during sterilization, procedures for recalling already distributed instruments are insufficient [10,24]. The RFID technology proposed in this study can automatically record and track the complete reprocessing history, addressing these issues effectively.
Previous research has consistently highlighted the limitations of manual recordkeeping, including spatial, searchability, and preservation issues, emphasizing the need for systematic sterilization documentation [25,26]. The RFID-based system proposed here automates instrument reprocessing records, reducing reliance on manual documentation, minimizing errors and omissions, and enabling rapid and accurate traceability in case of suspected infections. According to the World Health Organization (WHO), approximately 10% of hospitalized patients experience healthcare-associated infections (HAIs) [27]. Applying this system in clinical practice could prevent infections caused by improper instrument use and allow prompt response in suspected cases.
The core functionality of the proposed system automatically records the processing history of each instrument at every stage cleaning, sterilization, storage, and usage and stores the information in a database in chronological order. Each instrument is managed with a unique identification number, and scanning the tag attached to an instrument or set intuitively displays the current status, such as “In Use,” “Cleaning,” “Sterilized,” “In Storage,” or “Expired.” This enables medical staff and sterilization personnel to monitor instrument status in real time without manual recordkeeping. Automation via RFID reduces staff workload, ensures smooth instrument turnover, and enhances clinical efficiency.
The system also allows administrators to set user-specific access permissions and utilize statistical analysis features. Unlike paper records, which are cumbersome to process, data stored in the server can be analyzed for monthly, quarterly, or yearly trends, supporting resource allocation and management strategies for infection control.
However, the initial investment cost for implementing an RFID system cannot be overlooked. The expenses associated with RFID tags, readers, and server infrastructure are significant, and careful consideration is required for clinical application. Nevertheless, the adoption of an RFID system is expected to provide economic benefits in various aspects, including labor cost reduction, improved operational efficiency through decreased recording errors, and reduced social and economic costs associated with infection incidents.
This study has several limitations. First, it was limited to the performance evaluation of RFID tags (chips) through simulations conducted in a controlled laboratory environment, and therefore, additional quantitative metrics are required to support its application in actual clinical settings. Second, the system’s capability for evaluating the performance of equipment required for instrument reprocessing was minimal; future work should integrate Internet of Things (IoT) technology to extend functionality, including monitoring the physical status of equipment. Third, the evaluation was based on short-term experiments, and further assessment is needed to examine the durability and lifespan of the equipment under prolonged use.
In conclusion, the wireless ICT- and RFID-based dental instrument reprocessing management system proposed in this study addresses the limitations of traditional manual and paper-based management methods and demonstrates the potential for systematic management of cleaning and sterilization histories in dental clinical settings. RFID-based automated recording and traceability allow real-time monitoring of instrument status, providing a foundation for improving infection control and ensuring patient safety. Long-term operational data analysis and IoT-enabled feature expansion could further establish a digital infection control system tailored for dental healthcare environments.
Notes
Conflicts of Interest
None